Interested in learning more about Hyfe? Read about all of our news, announcements and updates here!

Comprehensive Chronic Cough Report reveals $15B opportunity. 100M patients, zero FDA-approved drugs, positioning digital cough monitoring as essential pharma infrastructure.

Review including Hyfe research evaluates continuous digital cough monitoring as a practical way to address limitations of patient-reported cough data

Study results demonstrate digital Behavioral Cough Suppression Therapy reduced cough frequency by over 40% in patients with refractory and unexplained chronic cough.
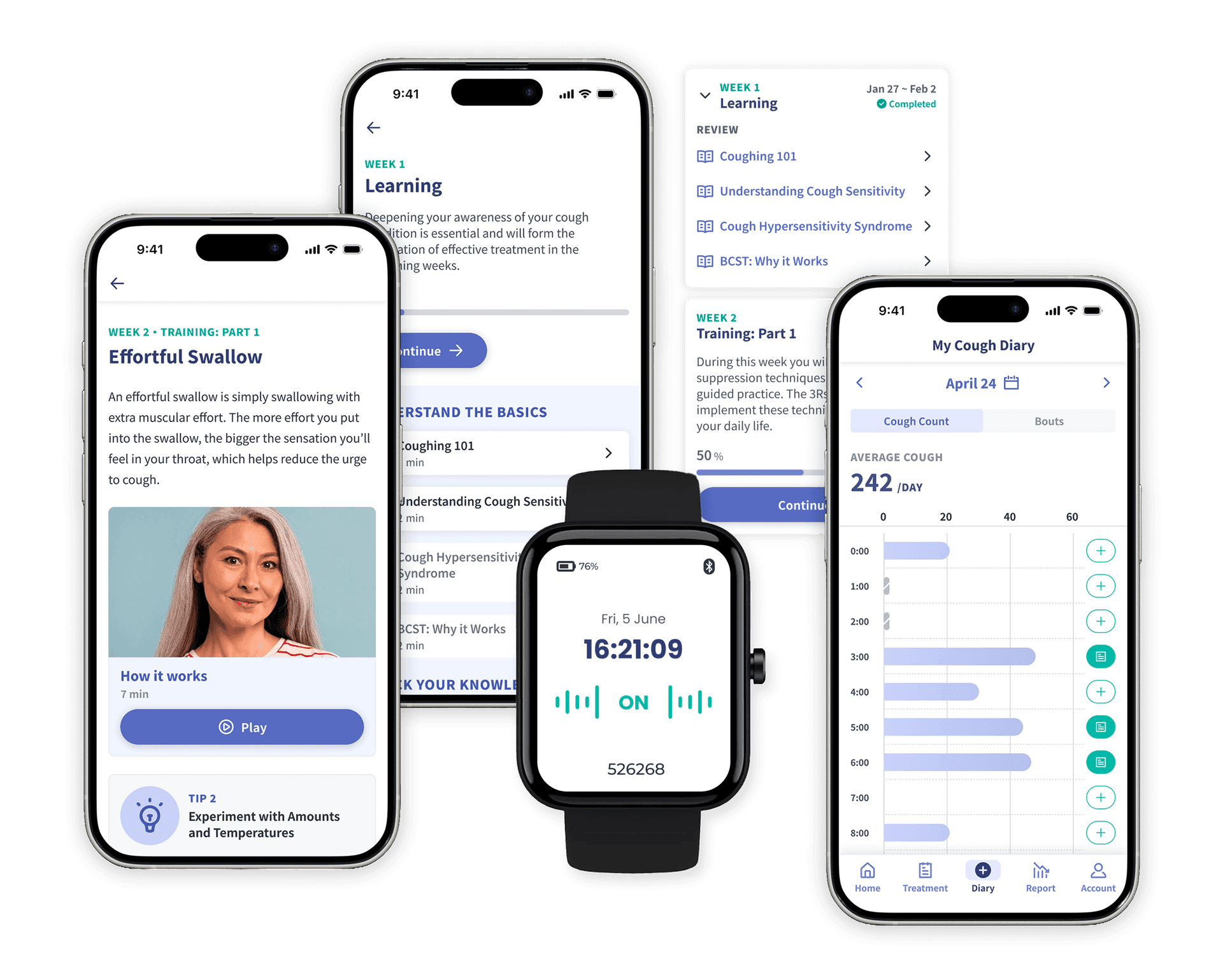
Strategic partnership with KYORIN Pharmaceuticals aims to improve outcomes and expand treatment access to millions impacted by chronic cough globally.
20.01.2026

Pipeline report documents 100M patient addressable market, first-mover economics, and digital therapeutic revenue impact of at least 44% lifetime value increase
Wilmington, Del., Jan 20, 2025 – Hyfe, Inc., the global leader in AI-powered cough care, released a new market intelligence report documenting a substantial commercial opportunity in Refractory Chronic Cough (RCC) therapeutics, where approximately 100 million patients worldwide and 10 million in the U.S. have no access to FDA-approved treatment options. The report, Refractory Chronic Cough Therapeutics: Pipeline Analysis & Competitive Landscape 2026-2035, provides the most detailed competitive assessment of the RCC treatment landscape available.
Currently, the standard of care for RCC patients relies entirely on off-label neuromodulators with 31-40% adverse event rates. This analysis projects the global cough therapeutic market will reach $14-15 billion by 2035 from approximately $9 billion in 2024, representing a 6% compound annual growth rate across the U.S., EU, U.K., and Japan. With major industry signals and regulatory outcomes, such as GSK’s acquisition of Bellus Health and Merck’s Complete Response Letters for Gefapixant, there is clear market validation and regulatory requirements driving the adoption of RCC therapeutics.
The 52-page analysis quantifies market dynamics across regulatory, clinical, and commercial dimensions, evaluating three primary therapeutic approaches including P2X3 Antagonists like Camlipixant, Kappa/Mu Opioid Modulators such as Trevi Therapeutics’ Nalbuphine ER, and Digital Therapeutics like the Kyorin Pharmaceuticals × Hyfe collaboration. It details:
The report concludes the U.S. market will support one, possibly two, meaningfully differentiated therapies based on: temporal separation between approvals, specialist referral bottlenecks limiting rapid market expansion, digital ecosystem lock-in effects once patients engage with monitoring platforms, and payer preference for a single preferred agent absent clear clinical differentiation. Durability of evidence, data integration, and health economics validation will determine market share allocation.
RCC is a widely underserved market, and what stands out in this report is how clearly defined the opportunity has become. We built this analysis to help development and commercial teams understand where differentiation is possible and how real-world care pathways are shaping which therapies can realistically succeed.
Dr. Peter Small
MD, Chief Medical Officer at Hyfe
The complete analysis, including competitive landscape matrices, payer reimbursement frameworks by jurisdiction, and detailed financial models, is available for free at coughreport.com. This research was conducted with financial support from Hyfe Inc. The views and conclusions expressed are those of the authors and do not necessarily reflect the views of Hyfe Inc.
Hyfe, Inc. is the global leader in AI-powered cough-monitoring technology. The company’s patented machine-learning software enables passive, long-term monitoring of cough frequency, revealing novel health insights. Hyfe delivers cough-monitoring solutions that empower pharma and academic researchers to advance clinical trials, enable healthcare providers to optimize patient care, and power digital therapeutics to transform the management of respiratory illness, including chronic cough. Hyfe’s technology has been used in 50+ research studies, and its long-term partners include global pharma companies and leading academic institutions. More information is available at hyfe.com.
###
Media contact:
press@hyfe.com